The 2022 Nobel Prize in Chemistry
Samaresh Jana
Department of Chemistry, School of Applied Sciences, KIIT-Deemed to be University, Bhubaneswar, Odisha
Noble Prize winners in Chemistry’ 2022

The 2022 Nobel Prize in Chemistry has been awarded to Prof. Carolyn R. Bertozzi, Prof. Morten Meldal and Prof. Karl Barry Sharpless, (see the picture above) for the development and extensive application of Click Chemistry and Bioorthogonal Chemistry. In this write-up we first write about the scientists and then about their discovery for which they were awarded the Nobel Prize.

Prof. Karl Barry Sharpless (Source: https://www.x-mol.com/university/faculty/689)
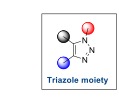
Professor Sharpless is an American chemist and is well known for his work on enantioselctive reactions. He was born in Philadelphia, US and earned Ph.D. in Organic Chemistry from Stanford University. He had served Stanford University and MIT as a Professor. Prof. Sharpless was awarded the Nobel Prize in Chemistry in 2001 for the development of enantioselctive epoxidation reaction. At that time, he was involved in developing an interesting chemical reaction which is called “Click Reaction”. Sharpless first introduced the term Click Chemistry in 2000. This is a special type of chemical reaction which follows certain conditions. If we think about various Click reactions, 1,3-dipolar cycloaddition reaction of azide (a nitrogenous compound) with an alkyne (a compound having triple bond) has been found to be the best reaction in the field of chemical sciences. His group discovered this transformation to produce a triazole moiety (a cyclic compound having three nitrogen atoms connected together) in an elegant way without generating any side product.

Prof. Morten Meldal (Source: https://en.wikipedia.org/wiki/Morten_P._Meldal)
Prof. Meldal serving at the University of Copenhagen, Denmark, had obtained Ph.D. in Chemistry from Technical University of Denmark (DTU) following which he carried out his postdoctoral work at DTU, Cambridge University and University of Copenhagen. In 1998, Prof. Meldal established a synthetic chemistry laboratory at the Department of Chemistry, Carlsberg Laboratory, Denmark. Prof. Meldal’s group has been doing research in the area of synthetic chemistry and developing technological methods and instruments for peptide synthesis. His group discovered the Copper Catalysed Azide Alkyne Cycloaddition (CuAAC) reaction during the same time when Sharpless reported the click reaction. Based on the developed chemistry, Prof. Meldal produced various kinds of useful materials such as medicinal compounds and biologically important materials.

Prof. C. R. Batorzzi (Source: https://en.wikipedia.org/wiki/Carolyn_Bertozzi)
Prof. C. R. Bertozzi, an American Chemist and Biologist received Ph. D. in Chemistry from the University of California, Barkley. After several assignments at various Universities of US, presently she is engaged at the Department of Chemistry, Stanford University. Her group contributed tremendously to Click Chemistry; in particular, they have devised a strategy to perform Click Chemistry on biomolecules. The group has been quite successful in implementing the reaction on the living system without hampering the other reactions of the living system.
Contributions behind the Award
To know the contribution of these three Nobel Laureates, let us understand their discovery in a simple way. In general, a chemical reaction is a process where we convert a starting material or a reactant into a useful product. But the problem is that in all chemical reactions, we can’t get our desired product easily or in a major amount. We have to control the reaction in a particular manner, so that we can obtain only our required product, not a side product. For this purpose, a scientist needs to design a substrate or catalyst and also needs to optimize the reaction conditions in a proper way. This is a challenging work. The three Nobel Laureates accomplished the challenging work in a novel way. They discovered a special type of chemical reaction which helps the human being a lot. Health is the best wealth of human being. In this era, because of the environmental pollution, lifestyle and food habit, humans suffer from a variety of diseases. We take medicines for making our lives much better. Most of the medicines contain heterocyclic chemicals, particularly nitrogenous compounds. The three Nobel Laureates worked on a topic of chemistry, which basically helps us to make medicinally important nitrogenous compounds. Their chemistry is related to the development of a transformation called “Click Reaction”. It is not a very specific type of chemical reaction, but if any reaction fulfils certain criteria such as modularity, insensitive to solvent, high chemical yield, regiospecificity and lower yield of side products, then the reaction is called a Click Reaction. Out of several reactions in Synthetic Organic Chemistry, 1,3-dipolar cycloaddition reaction is an example of Click reaction. It is believed to be a single-step reaction between a terminal alkyne (triple bond containing compound) and an azide (nitrogenous compound) moiety to form a triazole (see the diagram below). Triazole is a five membered cyclic compound and it contains three nitrogen atoms connected together as shown below. This type of compound can be used in medicinal chemistry for therapeutic use because the triazole moiety can interact with the living part in our body.
To make the triazole compound, scientist had tried various processes including dipolar cycloaddition (Huisgen cycloaddition) reaction but had failed to achieve the desired isomeric compound (particularly one regioisomer) in major amount.
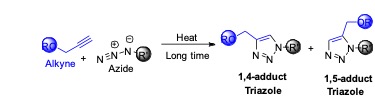
Scheme: Huisgen 1,3-dipolar cycloaddition reaction
Under heating condition for a long time, an azide reacts with an alkyne to produce 1,4- adduct and 1,5-adduct of triazole (see the scheme above). There is no selectivity in this reaction and the reaction time is quite high even at elevated temperature. For this reason, a development was required to make this reaction in a regioselective manner. Since 2000, Prof. Meldal and Prof. Sharpless simultaneously started work on the development on this chemical transformation using a copper(I) salt as a catalyst. Interestingly, it is observed that in the presence of copper catalyst, only 1,4-adduct of the triazole is obtained as a sole product. Mechanistic study reveals that the reaction is no longer a true concerted one. Therefore, it is not a 1,3-dipolar cycloaddition and should not be termed as Huisgen cycloaddition. Further, the reaction is named as Copper(I) catalysed Azide Alkyne Cycloaddition (CuAAC) or click reaction.
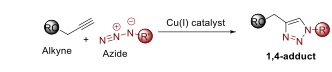
Copper(I) catalysed Azide Alkyne Cycloaddition (CuAAC).
Click reaction has potential application in various fields, for example in polymerization reaction, in material science, in surface science, etc. But its application in biomolecules or inside the living system makes this a novel work. However, due to the toxic nature of metal catalysts, copper-catalysed click reaction could not be performed inside the living system. Prof. Bertozzi tried the click chemistry on the living system and worked in this field for several decades without any metal catalyst. After huge efforts, her group developed a simple technique by which one can run a click reaction on the living system without any toxic metal catalysts. She took this click chemistry as a challenge and transferred the reaction in a new dimension. Thus, Copper catalyst-free Click reaction in biomolecules is referred to as bioorthogonal reaction coined by Prof. Bertozzi. She extensively applied this click chemistry in various living systems. Interestingly, the reaction does not affect any other cell reactions.
Bioorthogonal chemistry is an attractive technique in the field of nuclear imaging and radiotherapy. Moreover, it is also applied to cancer therapy. Utilization of click chemistry in diverse fields, particularly in the living system, makes the chemical reaction extremely novel.


